
#Two types of nodes occur in atomic orbitals full
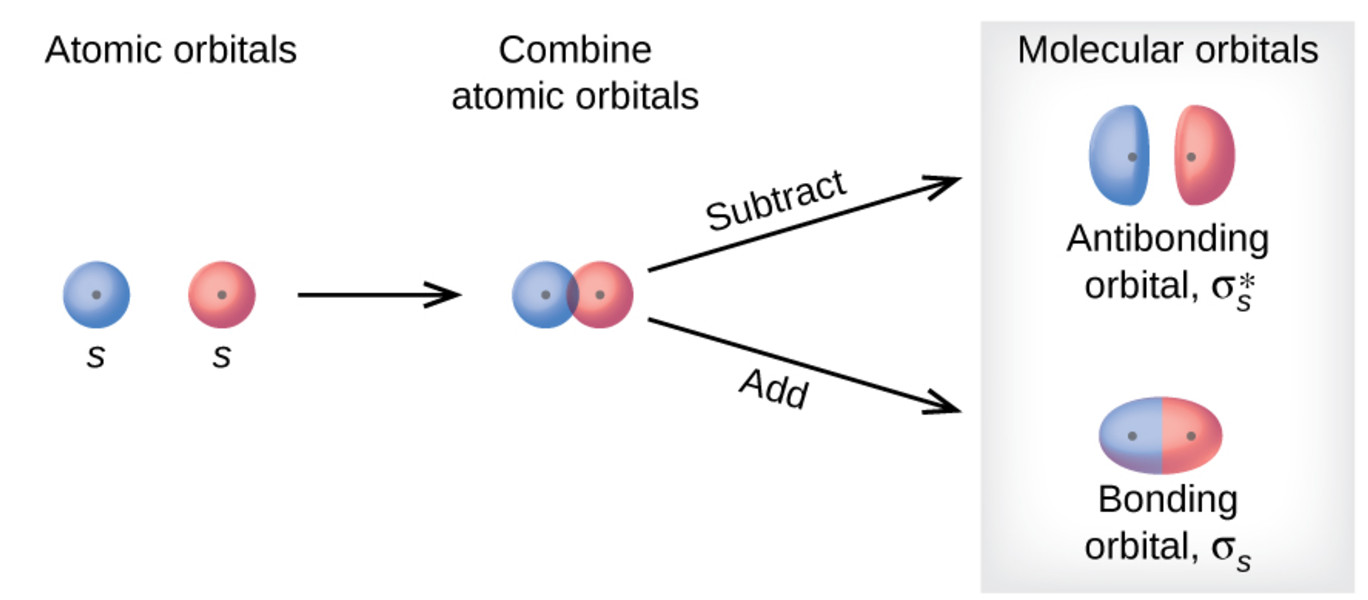
Like an atomic orbital, a molecular orbital is full when it contains two electrons with opposite spin. The region of space in which a valence electron in a molecule is likely to be found is called a molecular orbital ( Ψ 2). Just like electrons around isolated atoms, electrons around atoms in molecules are limited to discrete (quantized) energies. Using quantum mechanics, the behavior of an electron in a molecule is still described by a wave function, Ψ, analogous to the behavior in an atom. Molecular orbital theory describes the distribution of electrons in molecules in much the same way that the distribution of electrons in atoms is described using atomic orbitals. Needs multiple structures to describe resonance Predicts the arrangement of electrons in molecules Predicts molecular shape based on the number of regions of electron density Comparison of Bonding TheoriesĬonsiders bonds as localized between one pair of atomsĬonsiders electrons delocalized throughout the entire moleculeĬreates bonds from overlap of atomic orbitals ( s, p, d…) and hybrid orbitals ( sp, sp 2, sp 3…)Ĭombines atomic orbitals to form molecular orbitals (σ, σ*, π, π*)Ĭreates bonding and antibonding interactions based on which orbitals are filled Both theories provide different, useful ways of describing molecular structure. Table 1 summarizes the main points of the two complementary bonding theories. MO theory also helps us understand why some substances are electrical conductors, others are semiconductors, and still others are insulators. Unlike valence bond theory, which uses hybrid orbitals that are assigned to one specific atom, MO theory uses the combination of atomic orbitals to yield molecular orbitals that are delocalized over the entire molecule rather than being localized on its constituent atoms. Additionally, it provides a model for describing the energies of electrons in a molecule and the probable location of these electrons. It also explains the bonding in a number of other molecules, such as violations of the octet rule and more molecules with more complicated bonding (beyond the scope of this text) that are difficult to describe with Lewis structures. Molecular orbital theory (MO theory) provides an explanation of chemical bonding that accounts for the paramagnetism of the oxygen molecule. You can see videos of diamagnetic floating frogs, strawberries, and more at the Radboud University Diamagnetic Levitation website. If you place a frog near a sufficiently large magnet, it will levitate. Living things contain a large percentage of water, so they demonstrate diamagnetic behavior. Water, like most molecules, contains all paired electrons.
